
动物实验低压氧舱


动物实验低压氧舱用于模拟低压氧高原环境,压力可以根据需要自行设置,最高可模拟海拔高度12000米,适用于更多的鼠笼或中小型动物如犬、猴、兔、鼠等。
整个实验装置由动物舱体、真空系统、监测及控制系统构成。设备的自动化程度高,无须专人守候,可长期持续一个月运行。
产品特点:
人性化操作
采用10英寸触摸屏,人性化界面设计,操作简便。
两种控制方式:自动、手动,断电可操控;具有传感器组件及机械表双重监测功能。设备自动化程序设计,无须专人守候,可长期持续运转。
实时显示海拔和氧分压动态变化曲线。
实验过程数据可导出csv格式保存到U盘中,在电脑中读取分析。
可根据客户实验需求,订制实验环境及配套其他相关实验设备。
配备动物低压专用水瓶,防止在负压环境中,水滴漏速过快。
操作安全放心
配备自动换气功能,符合动物饲养规范。
提供多方位报警功能(温度、湿度、压力、氧浓度 上/下限),防止出现安全隐患。配备渐开手轮式舱门,降低因为误操作造成的伤害。
舱体采用透明厚亚克力材质,方便研究人员观察实验动物情况。
配备气体缓冲器,减小、甚至消除了细股急流气体对小动物的影响。
配备多级空气过滤器,确保动物舱内实验环境空气不会收到污染。
参数显示/功能全
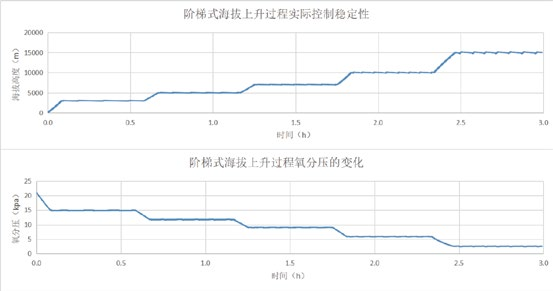

拓展及特殊定制
动物生理指标监测
可实现的监测指标:心电图、心率、体温、血压、呼吸、血氧饱和度;
呼吸代谢监控功能
采血给药功能
视频监测功能
动物低氧跑台装置
低氧强迫游泳装置
温控功能
恒温功能,温度可控制,20~45℃;
低温功能,4~45℃
可定制其它功能
应用领域
高原医学研究、肺水肿、脑水肿、肺动脉高压等疾病研究
型号说明
|
名称 |
型号 |
说明 |
|
动物低压氧舱 (标准版) |
ProOx-810 |
可以摆放2个大鼠笼/4个小鼠笼 |
|
动物低压氧舱 (温控版) |
ProOx-811 |
可以摆放2个大鼠笼/4个小鼠笼,同时加上温控功能,标配:20~45℃;可选配:4~45℃ |
|
动物低压氧舱 (大容量版) |
ProOx-810L |
可以摆放4个大鼠笼/8个小鼠笼(可叠加) |
|
动物低压氧舱 (大容量版+温控) |
ProOx-811L |
可以摆放4个大鼠笼/8个小鼠笼(可叠加),同时加上温控功能,标配:20~45℃;可选配:4~45℃ |
|
动物低压氧舱 基础款 |
ProOx-830 |
可以摆放1个大鼠笼/2个小鼠笼 |
|
动物低压氧舱 (加大容量版) |
ProOx-810L-L |
可以摆放6个大鼠笼/12个小鼠笼(可叠加) |
|
动物低压氧舱 (加大容量版+温控) |
ProOx-811L-L |
可以摆放6个大鼠笼/12个小鼠笼(可叠加),同时加上温控功能,标配:20~45℃;可选配:4~45℃ |
客户名单(部分)
相关文献
[1] Drekolia M K, Mettner J, Wang D, et al. Cystine import and oxidative catabolism fuel vascular growth and repair via nutrient-responsive histone acetylation[J]. Cell Metabolism (IF 30.9), 2025.
[2] Wu L W, Chen M, Jiang C Y, et al. Inactivation of AXL in Cardiac Fibroblasts Alleviates Right Ventricular Remodeling in Pulmonary Hypertension[J]. Advanced Science (IF 14.1), 2025: e08995.
[3] Lei R, Gu M, Li J, et al. Lipoic acid/trometamol assembled hydrogel as injectable bandage for hypoxic wound healing at high altitude[J]. Chemical Engineering Journal (IF 13.4), 2024, 489: 151499.
[4] Li Z, Li H, Qiao W, et al. Multi-omics dissection of high TWAS-active endothelial pathogenesis in pulmonary arterial hypertension: bridging single-cell heterogeneity, machine learning-driven biomarkers, and developmental reprogramming[J]. International Journal of Surgery (IF 10.1), 10.1097.
[5] Pei Y, Huang L, Wang T, et al. Bone marrow mesenchymal stem cells loaded into hydrogel/nanofiber composite scaffolds ameliorate ischemic brain injury[J]. Materials Today Advances (IF 10), 2023, 17: 100349.
[6] Wang Q, Liu J, Li R, et al. Macrophage κ-opioid receptor inhibits hypoxic pulmonary hypertension progression and right heart dysfunction via an SCD1-dependent anti-inflammatory response[J]. Genes & Diseases (IF 9.4), 2025: 101604.
[7] Wang Y, Zhang R, Chen Q, et al. PPARγ Agonist Pioglitazone Prevents Hypoxia-induced Cardiac Dysfunction by Reprogramming Glucose Metabolism[J]. International Journal of Biological Sciences, 2024, 20(11): 4297.
[8] Wang Y, Shen P, Wu Z, et al. Plasma Proteomic Profiling Reveals ITGA2B as a key regulator of heart health in high-altitude settlers[J]. Genomics, Proteomics & Bioinformatics, 2025: qzaf030.
[9] Lan Y, Zhao S, Song Y, et al. Physicochemical properties of selenized quinoa protein hydrolysate and its regulatory effects on neuroinflammation and gut microbiota in hypoxic mice[J]. Journal of Future Foods, 2025.
[10] Pan Z, Yao Y, Liu X, et al. Nr1d1 inhibition mitigates intermittent hypoxia-induced pulmonary hypertension via Dusp1-mediated Erk1/2 deactivation and mitochondrial fission attenuation[J]. Cell Death Discovery, 2024, 10(1): 459.
[11] Zhou Y, Ni Z, Liu J, et al. Gut Microbiota‐Associated Metabolites Affected the Susceptibility to Heart Health Abnormality in Young Migrants at High‐Altitude: Gut Microbiota and Associated Metabolites Impart Heart Health in Plateau[C]//Exploration. 2025: 20240332.
[12] Li C, Zhao Z, Jin J, et al. NLRP3-GSDMD-dependent IL-1β Secretion from Microglia Mediates Learning and Memory Impairment in a Chronic Intermittent Hypoxia-induced Mouse Model[J]. Neuroscience, 2024, 539: 51-65.
[13] Yang W, Li M, Ding J, et al. High-altitude hypoxia exposure inhibits erythrophagocytosis by inducing macrophage ferroptosis in the spleen[J]. Elife, 2024, 12: RP87496.
[14] You Z, Huang Q, Zeng L, et al. Rab26 promotes hypoxia-induced hyperproliferation of PASMCs by modulating the AT1R-STAT3-YAP axis[J]. Cellular and Molecular Life Sciences, 2025, 82(1): 1-16.
[15] Pei C, Shen Z, Wu Y, et al. Eleutheroside B Pretreatment Attenuates Hypobaric Hypoxia‐Induced High‐Altitude Pulmonary Edema by Regulating Autophagic Flux via the AMPK/mTOR Pathway[J]. Phytotherapy Research, 2024, 38(12): 5657-5671.
[16] Duan H, Han Y, Zhang H, et al. Eleutheroside B Ameliorates Cardiomyocytes Necroptosis in High-Altitude-Induced Myocardial Injury via Nrf2/HO-1 Signaling Pathway[J]. Antioxidants, 2025, 14(2): 190.
[17] Song J, Zheng J, Li Z, et al. Sulfur dioxide inhibits mast cell degranulation by sulphenylation of galectin-9 at cysteine 74[J]. Frontiers in Immunology, 2024, 15: 1369326.
[18] Jia N, Shen Z, Zhao S, et al. Eleutheroside E from pre-treatment of Acanthopanax senticosus (Rupr. etMaxim.) Harms ameliorates high-altitude-induced heart injury by regulating NLRP3 inflammasome-mediated pyroptosis via NLRP3/caspase-1 pathway[J]. International Immunopharmacology, 2023, 121: 110423.
[19] Huang Q, Han X, Li J, et al. Intranasal Administration of Acetaminophen-Loaded Poly (lactic-co-glycolic acid) Nanoparticles Increases Pain Threshold in Mice Rapidly Entering High Altitudes[J]. Pharmaceutics, 2025, 17(3): 341.
[20] Wu Y, Tang Z, Du S, et al. Oral quercetin nanoparticles in hydrogel microspheres alleviate high-altitude sleep disturbance based on the gut-brain axis[J]. International Journal of Pharmaceutics, 2024, 658: 124225.
[21] Zhou Z, Zhao Q, Huang Y, et al. Berberine ameliorates chronic intermittent hypoxia‐induced cardiac remodelling by preserving mitochondrial function, role of SIRT6 signalling[J]. Journal of Cellular and Molecular Medicine, 2024, 28(12): e18407.
[22] Shang W, Huang Y, Xu Z, et al. The impact of a high-carbohydrate diet on the cognitive behavior of mice in a low-pressure, low-oxygen environment[J]. Food & Function, 2025, 16(3): 1116-1129.
[23] Pei C, Jia N, Wang Y, et al. Notoginsenoside R1 protects against hypobaric hypoxia-induced high-altitude pulmonary edema by inhibiting apoptosis via ERK1/2-P90rsk-BAD ignaling pathway[J]. European Journal of Pharmacology, 2023, 959: 176065.
[24] Xie L, Wu Q, Huang H, et al. Neuroregulation of histamine of circadian rhythm disorder induced by chronic intermittent hypoxia[J]. European Journal of Pharmacology, 2025: 177662.
[25] Ding Y, Liu W, Zhang X, et al. Bicarbonate-Rich Mineral Water Mitigates Hypoxia-Induced Osteoporosis in Mice via Gut Microbiota and Metabolic Pathway Regulation[J]. Nutrients, 2025, 17(6): 998.
[26] Gu N, Shen Y, He Y, et al. Loss of m6A demethylase ALKBH5 alleviates hypoxia-induced pulmonary arterial hypertension via inhibiting Cyp1a1 mRNA decay[J]. Journal of Molecular and Cellular Cardiology, 2024.
[27] Luan X, Zhu D, Hao Y, et al. Qibai Pingfei Capsule ameliorated inflammation in chronic obstructive pulmonary disease (COPD) via HIF-1 α/glycolysis pathway mediated of BMAL1[J]. International Immunopharmacology, 2025, 144: 113636.
[28] Jiang H, Lu C, Wu H, et al. Decreased cold‐inducible RNA‐binding protein (CIRP) binding to GluRl on neuronal membranes mediates memory impairment resulting from prolonged hypobaric hypoxia exposure[J]. CNS Neuroscience & Therapeutics, 2024, 30(9): e70059.
[29] Chang P, Xu M, Zhu J, et al. Pharmacological Inhibition of Mitochondrial Division Attenuates Simulated High‐Altitude Exposure‐Induced Memory Impairment in Mice: [30] Involvement of Inhibition of Microglia‐Mediated Synapse Elimination[J]. CNS Neuroscience & Therapeutics, 2025, 31(6): e70473.
[30] Liu C, Qu D, Li C, et al. miR‐448‐3p/miR‐1264‐3p Participates in Intermittent Hypoxic Response in Hippocampus by Regulating Fam76b/hnRNPA2B1[J]. CNS Neuroscience & Therapeutics, 2025, 31(2): e70239.
[31] Wu L W, Chen M, Jiang D J, et al. TCF7 enhances pulmonary hypertension by boosting stressed natural killer cells and their interaction with pulmonary arterial smooth muscle cells[J]. Respiratory Research, 2025, 26(1): 202.
[32] Xie L, Wu Q, Huang H, et al. Neuroregulation of histamine of circadian rhythm disorder induced by chronic intermittent hypoxia[J]. European Journal of Pharmacology, 2025: 177662.
[33] Cai S, Li Z, Bai J, et al. Optimized oxygen therapy improves sleep deprivation-induced cardiac dysfunction through gut microbiota[J]. Frontiers in Cellular and Infection Microbiology, 2025, 15: 1522431.
[34] Wang X, Xie Y, Niu Y, et al. CX3CL1/CX3CR1 signal mediates M1-type microglia and accelerates high-altitude-induced forgetting[J]. Frontiers in Cellular Neuroscience, 2023, 17: 1189348.
[35] He Y, Wang Y, Duan H, et al. Pharmacological targeting of ferroptosis in hypoxia-induced pulmonary edema: therapeutic potential of ginsenoside Rg3 through activation of the PI3K/AKT pathway[J]. Frontiers in Pharmacology, 2025, 16: 1644436.
[36] Guo Y, Qin J, Sun R, et al. Molecular hydrogen promotes retinal vascular regeneration and attenuates neovascularization and neuroglial dysfunction in oxygen-induced retinopathy mice[J]. Biological Research, 2024, 57.
[37] Liu L, Zhang J, Song S, et al. Paraventricular nucleus neurons: important regulators of respiratory movement in mice with chronic intermittent hypoxia[J]. Annals of Medicine, 2025, 57(1): 2588664.
[38] Ma Q, Ma J, Cui J, et al. Oxygen enrichment protects against intestinal damage and gut microbiota disturbance in rats exposed to acute high-altitude hypoxia[J]. Frontiers in Microbiology, 2023, 14.
[39] Lan J, Lin J, Guo Y, et al. Sequencing and bioinformatics analysis of exosome-derived miRNAs in mouse models of pancreatic injury induced by OSA[J]. Frontiers in Physiology, 2025, 16: 1712442.
[40] Feng X, Li C, Zhang W, et al. Mechanism of retinal angiogenesis induced by HIF-1α and HIF-2α under hyperoxic conditions[J]. Scientific Reports, 2025, 15(1): 36049.
[41] Yao Y, Chen Y, Li Y, et al. TGM2 Enhances Hypobaric Hypoxia-mediated Brain Injury Via Regulating NLRP3/GSDMD Signaling[J]. Neurochemical Research, 2025, 50(6): 1-11.
[42] Yang A, Guo L, Zhang Y, et al. MFN2-mediated mitochondrial fusion facilitates acute hypobaric hypoxia-induced cardiac dysfunction by increasing glucose catabolism and ROS production[J]. Biochimica et Biophysica Acta (BBA)-General Subjects, 2023: 130413.
[43] Chu H, Jiang W, Zuo N, et al. Astrocyte activation: A key mediator underlying chronic intermittent hypoxia-induced cognitive dysfunction[J]. Sleep Medicine, 2025: 106692.
[44] Xu A, Huang F, Chen E, et al. Hyperbaric oxygen therapy attenuates heatstroke-induced hippocampal injury by inhibiting microglial pyroptosis[J]. International Journal of Hyperthermia, 2024, 41(1): 2382162.
[45] Zhang Z, Zheng X, He Y, et al. Hyperbaric oxygen ameliorates neuroinflammation in heat-stressed BV-2 microglial cells: potential involvement of EAAT2 regulation[J]. International Journal of Hyperthermia, 2025, 42(1): 2583133.
[46] Jinyu F, Huaicun L, Yanfei Z, et al. Nogo-A Protein Mediates Oxidative Stress and Synaptic Damage Induced by High-altitude Hypoxia in the Rat Hippocampus[J]. 2024.
[47] Su L, Ni T, Fan R, et al. An attention to the effect of intravitreal injection on the controls of oxygen-induced retinopathy mouse model[J]. Experimental Eye Research, 2024, 248: 110094.
[48] Xu Y, Xu J, Li J, et al. Interplay of HIF-1α, SMAD2, and VEGF signaling in hypoxic renal environments: impact on macrophage polarization and renoprotection[J]. Renal Failure, 2025, 47(1): 2561784.
[49] Zhang D, Bian W, Gao Z. Impact of Obstructive Sleep Apnea on Endometrial Function in Female Rats: Mechanism Exploration[J]. Nature and Science of Sleep, 2025: 2485-2499.
[50] Zhang N, Wei F, Ning S, et al. PPARγ Agonist Rosiglitazone and Antagonist GW9662: Antihypertensive Effects on Chronic Intermittent Hypoxia-Induced Hypertension in Rats[J]. Journal of Cardiovascular Translational Research, 2024: 1-13.
[51] Zhang Y, Zhang A, Yang J, et al. Hypoxic Mesenchymal Stem Cell Exosome‐Derived SLC25A3 Ameliorates Bronchopulmonary Dysplasia by Modulating Macrophage Polarization and Oxidative Stress[J]. Cell Biochemistry and Function, 2025, 43(12): e70152.
[52] Lan J, Wang Y, Liu C, et al. Genome-wide analysis of m6A-modified circRNAs in the mouse model of myocardial injury induced by obstructive sleep apnea[J]. BMC Pulmonary Medicine, 2025, 25(1): 158.
[53] Zhang L, Liu X, Wei Q, et al. Arginine attenuates chronic mountain sickness in rats via microRNA-144-5p[J]. Mammalian Genome, 2023, 34(1): 76-89.
[54] Wei J, Hu M, Chen X, et al. Hypobaric Hypoxia Aggravates Renal Injury by Inducing the Formation of Neutrophil Extracellular Traps through the NF-κB Signaling Pathway[J]. Current Medical Science, 2023: 1-9.
[55] Zhang L, Li J, Wan Q, et al. Intestinal stem cell-derived extracellular vesicles ameliorate necrotizing enterocolitis injury[J]. Molecular and Cellular Probes, 2025, 79: 101997.
[56] Liao Y, Ke B, Long X, et al. Abnormalities in the SIRT1-SIRT3 axis promote myocardial ischemia-reperfusion injury through ferroptosis caused by silencing the PINK1/Parkin signaling pathway[J]. BMC Cardiovascular Disorders, 2023, 23(1): 582.
[57] Wang M, Wen W, Chen Y, et al. TRPC5 channel participates in myocardial injury in chronic intermittent hypoxia[J]. Clinics, 2024, 79: 100368.
[58] Li J, Ye J. Chronic intermittent hypoxia induces cognitive impairment in Alzheimer’s disease mouse model via postsynaptic mechanisms[J]. Sleep and Breathing, 2024: 1-9.
[59] Binbin L I, Haizhen L I, Houhuang C, et al. Utilizing Hyperbaric Oxygen Therapy to Improve Cognitive Function in Patients With Alzheimer’s Disease by Activating Autophagy-Related Signaling Pathways[J]. Physiological Research, 2025, 74(1): 141.
[60] Han J, Wang L, Wang L, et al. 5-Hydroxytryptamine Limits Pulmonary Arterial Hypertension Progression by Regulating Th17/Treg Balance[J]. Biological and Pharmaceutical Bulletin, 2025, 48(5): 555-562.
[61] Nan L, Kaisi F, Mengzhen Z, et al. miR-375-3p targets YWHAB to attenuate intestine injury in neonatal necrotizing enterocolitis[J]. Pediatric Surgery International, 2024, 40(1): 63.
[62] Liu B, Zheng W, Tang C, et al. Scutellarein-containing novel formula attenuates hypoxia through inhibiting apoptosis[J]. 2025.
参考研究
高原疾病介绍
不同的海拔高度大气压和氧分压的变化对比
*我公司可提供3Q验证,根据客户的特殊应用、特殊需求提供功能定制服务,也可以提供相关的实验服务,详情请来电咨询。

上海塔望智能科技有限公司响应“中国制造2025”的战略号召,2018年成立于上海交科科创园内,专注于临床前动物实验相关设备的开发制造。致力于在该领域打破国外产品垄断,树立起值得骄傲的中国制造品牌。
公司目前的产品线包括:吸入式毒理、动物呼吸肺功能检测、气体浓度控制、能量代谢系统、激光散斑血流成像系统等,所有产品均为自主研发,打破了长期以来进口产品的垄断,部分产品性能已超过进口产品。同时塔望科技融合生命科学、医学、IT、电子、机械等领域先进技术,为客户提供量身定制方案,满足个性化的需求。
我们在生命科学、医药研发等领域也在迅速追赶、逐渐超越欧美国家。目前落后于西方的生命科学仪器设备研发制造业也必然在中国创新创造的大背景下,迎来新的超越。这是时代给我们的机会。匠心筑梦,诚信筑塔,严谨求实,展望将来。塔望科技坚守工匠精神,用心做好每一件产品。我们将在这个瞬息万变的黄金时代,为本领域科学家提供值得骄傲和信赖的产品。




| 单位名称: |
|
详细地址:
上海市松江区泗砖路351号上海交科松江科创园
|
|
qq:
15221725700
|
| 联系电话: |
| Email: |



